Toxicity NH4+ in the plant ( Pampered little Musang King )
This is a typical problem of Musang king, “leaves burning” …
After plants absorb nutrients, they may not immediately use them. Before plants use these nutrients, they will be deposited in the cells of the leaves. If they do not use the nutrients immediately, they will begin to accumulate in these cells, nitrate-N (NO3-) can be safely stored in plant cells at high levels, but the accumulation of ammonium-N (NH4+) can damage plant cells.
Nitrogen is assimilated in both roots and leaves of crops (Marschner, 1995). When absorbing nitrate, 70% to 90% of nitrogen is transported to plant leaves in the form of nitrate (van Beusichem et al., 1988). In plant leaves, nitrate nitrogen is converted to ammonia. The toxic effects of ammonia on plant leaves are prevented when nitrogen combines with sugars in leaves to produce amino acids (usually glutamate) (Marschner, 1995), and the plant cells where sugars are produced are very close to being poisoned by ammonia. However, when ammonium enters the crop root system, all ammonium nitrogen is utilized by the metabolism of the crop root system, and the consumed sugar is transported to the crop root by phloem flow (Marschner, 1995). In crop roots, sugar serves 2 purposes: (i) cellular respiration and (ii) ammonium metabolism. However, when the temperature of the roots increased, the cellular respiration of the roots was enhanced, and the sugar concentration of the roots decreased. When the concentration of sugar in plant roots is reduced to an insufficient level for ammonium metabolism, free ammonia accumulates in root cells and poisoning root respiration , followed by plant root death (Ganmore-Newman and Kafkafi, 1985).
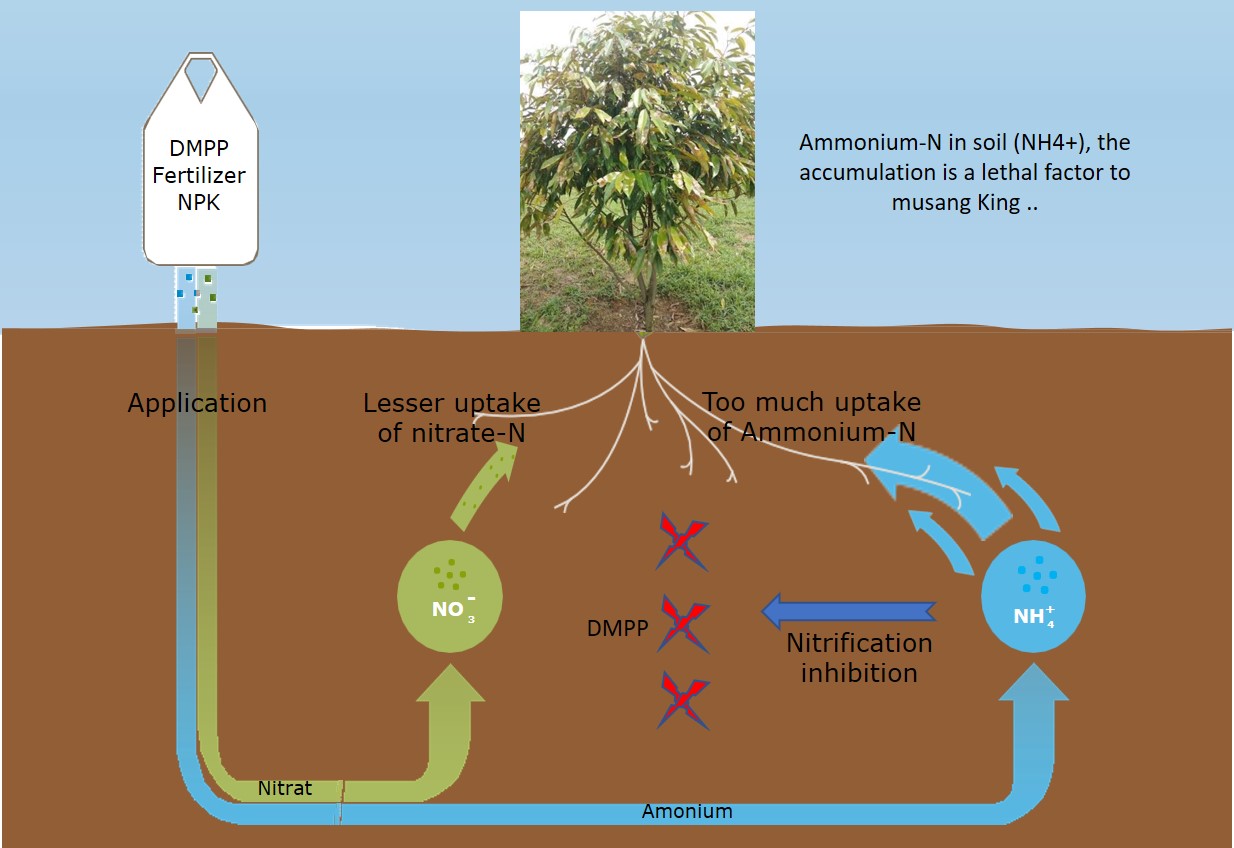
Many agricultural companies introduce high-efficiency fertilizers by adding nitrification inhibitors (dimethylpyrazole phosphate, DMPP). Nitrification inhibitors refer to chemicals that can inhibit the biological conversion process of ammonium-N into nitrate-N. They can selectively inhibit the activity of nitrifying bacteria in the soil, thereby retarding the reaction speed of ammonium-N in the soil into nitrate-N. Ammonium-N can be absorbed by soil colloids and is not easy to leach.
DMPP reduces N leaching for a period of 4 to 10 weeks, depending on soil temperature and soil humidity, the transformation of ammonium to nitrate is delayed. N availability is further adapted to the plants’ requirements. But, however, ammonium-N in soil (NH4+), the accumulation is a lethal factor, killing many crops in the forest. As far as I am concerned, nitrification inhibitors (equal to suicide) are not suitable for durian fertilizer management programs.
NH4+ is not only an important nitrogen source necessary for plant growth, but also a wide range of intermediate products existing in plant metabolism. However, when the external NH4+ concentration is too high, it can inhibit the growth of most higher plants. In this article, we discussed the susceptibility of different species to NH4+, and the symptoms of NH4+ poisoned plants, especially the mechanism of NH4+ poisoning and the methods to alleviate NH4+ poisoning. Different plants have different sensitivity to NH4+ poisoning. The growth of plants damaged by NH4+ poisoning is significantly inhibited. However, applying mixed nitrogen sources and increasing the K+ concentration in the culture conditions can reduce the degree of NH4+ poisoning.
 NH4+ in plants can be directly absorbed from the soil through roots, and it is also the product of deamination of some organic compounds in plants during certain pathways such as protein degradation, photorespiration, and biosynthesis of lignin. High concentration of NH4+ can inhibit the growth of plants. In some land, the concentration of NH4+ can reach 40 mmol/Lt (720ppm). When NH4+ is the only nitrogen source, it can produce NH4+ toxicity to plants. In addition, in the soil using urea as fertilizer, due to the action of bacterial urease, urea is converted into NH4+ and CO2 in a large amount, which greatly increases the NH4+ concentration in the soil, which can cause NH4+ poisoning of plants. In addition, many abiotic stresses can also cause the hyperaccumulation of NH4+ in plants. If NH4+ cannot be assimilated effectively, it can also produce NH4+ poisoning symptoms. And recent studies have found that the accumulation of excessive NH4+ is considered a factor in the extinction of plant species in various ecosystems. Therefore, the in-depth understanding of the NH4+ toxicity mechanism and the exploration of methods to alleviate the NH4+ toxicity are of great significance.
NH4+ in plants can be directly absorbed from the soil through roots, and it is also the product of deamination of some organic compounds in plants during certain pathways such as protein degradation, photorespiration, and biosynthesis of lignin. High concentration of NH4+ can inhibit the growth of plants. In some land, the concentration of NH4+ can reach 40 mmol/Lt (720ppm). When NH4+ is the only nitrogen source, it can produce NH4+ toxicity to plants. In addition, in the soil using urea as fertilizer, due to the action of bacterial urease, urea is converted into NH4+ and CO2 in a large amount, which greatly increases the NH4+ concentration in the soil, which can cause NH4+ poisoning of plants. In addition, many abiotic stresses can also cause the hyperaccumulation of NH4+ in plants. If NH4+ cannot be assimilated effectively, it can also produce NH4+ poisoning symptoms. And recent studies have found that the accumulation of excessive NH4+ is considered a factor in the extinction of plant species in various ecosystems. Therefore, the in-depth understanding of the NH4+ toxicity mechanism and the exploration of methods to alleviate the NH4+ toxicity are of great significance.
1 Classification of NH4+ sensitive species and NH4+ resistant species
The phenomenon of NH4+ poisoning is very common, but different plant species have different thresholds for responding to it. In mature plants, the symptoms of ammonium poisoning usually appear first on the lower old leaves, while the young plants appear first on the new leaves. Domesticated plants that are sensitive to NH4+ ion toxicity include tomatoes, potatoes, barley, peas, castor, mustard, beets, strawberries and citrus. In the soil of many natural ecosystems, NH4+ has increasingly become the main form of N source. Wild-type herbaceous plants Arnica and thistle parsley and Cistanche cistanche are also particularly sensitive to NH4+ toxicity.
Existing plants that are more resistant to NH4+ toxicity include rice and onions among domesticated species. Wild plants include heather, Ulagrass, Proteas and some temperate quilt trees. Even species that are highly resistant to NH4+ toxicity, when a sufficient amount of NH4+ is applied to them, they can also show toxic properties. For example, under the treatment of excessive NH4+, rice showed a phenotype of yellowing leaves and inhibited growth, especially under low potassium conditions. The deposition of a high degree of NH4+ can cause a large number of deaths of the red spruce in the forest, which is considered to be highly resistant to NH4+. The resistance to NH4+ poisoning varies with the species and the growth period.
NH4+ poisoning can change the morphology of plants. The concentration of exogenous NH4+ that can produce NH4+ poisoning is generally 0.1~0.5mmol/L. Barley is a plant that is sensitive to NH4+. Studies on it have shown that NH4+ toxicity can cause yellowing of barley leaves and inhibit plant growth, especially root elongation. In addition, NH4+ poisoning can also cause other visible symptoms, such as lowering the root/leaf ratio and reducing yield. More importantly, NH4+ poisoning can also inhibit the germination and seedling establishment of seeds, and it can cause the extinction of certain seed plants in nature. The accumulation of excessive NH4+ in plants can also affect the absorption of certain nutrient elements and the balance of hormones, and can lead to a decrease in the concentration of soluble carbonic acid. At the same time, it can increase the concentration of amino acids. In addition, NH4+ stress can also induce a large amount of ROS (reactive oxygen species) in plants, including superoxide radicals O2- and H2O2. Although the absorption of inorganic cations is reduced under NH4+ poisoning conditions, the total amount of NH4+ absorbed is still very large, resulting in that the cation concentration in the plant is still higher than the anion concentration. At the same time, plants poisoned by NH4+ can acidify the environment around the root system. The possible reason for this phenomenon is to balance the charge disorder in the plant, and a large number of protons flow out from the plant. However, plants cultivated under nitrate nitrogen conditions can cause alkalization of the surrounding environment.
 3 Mechanism of excessive NH4+ toxicity
3 Mechanism of excessive NH4+ toxicity
Many researchers have speculated on the principle of the phenomenon that excessive NH4+ can be harmful to plants. Most people believe that a large amount of NH4+ in plants leads to a decrease in the absorption of K+, Mg2+ and Ca2+, thereby causing ion disorders in plants. The rapid assimilation of NH4+ in roots requires a large amount of carbon skeleton as a substrate to complete the assimilation of NH4+ into amino acids This process leads to insufficient carbon skeletons in the roots of plants, and eventually causes toxicity. The assimilation of NH4+ can also lead to the release of H+, and further cause decarboxylation reaction, which is a decrease in the concentration of carbonic acid in the cell. The decrease in the concentration of carbonic acid caused by the decarboxylation of the root further leads to the lack of the carbon skeleton in the root, which causes an increase in the absorption of anions to balance the intracellular charge. Some researchers believe that NH4+ toxicity is related to the hormone disorder in plants. The phenomenon of plant main root growth inhibition caused by excessive NH4+ is believed to be related to auxin transport or signal pathways, and ethylene production is found in the leaves of plants with higher NH4+ concentration. In addition, some researchers believe that NH4+ toxicity is related to the reduction of light sum rate. In addition, some researchers believe that NH4+ toxicity is related to the decrease of light sum rate. The results of recent research on barley show that under high NH4+ concentration, the growth of barley is inhibited due to the waste of energy caused by NH4+ passing through the plasma membrane. Excessive NH4+ in the cell is transported out of the cell by an unknown transporter at the expense of energy. The high respiration rate in the roots and the energy requirement for NH4+ excretion suggest that this may be the essential reason for the damage of high concentrations of exogenous NH4+ to plants. In plant cells, excessive accumulation of NH4+ results in excessive transport of NH4+ across the plasma membrane, thereby forming NH4+ toxicity. Many bacteria in the environment of low concentration of K+ and high concentration of NH4+ ions, due to the transport of excess NH4+ across the plasma membrane, resulting in enhanced respiration. The latest research found that GDP-mannose pyrophosphorylase (GMPase) is related to NH4+ sensitivity in Arabidopsis roots. In this study, the decrease in GMPase activity caused defects in the N-glycosylation process of proteins, which is considered to be an important molecular level downstream step involved in the phenomenon of NH4+ inhibiting root growth. N-glycan activation is necessary for the correct folding of proteins, and plays an important role in initiating protein folding, cellulose synthesis, cell wall stability and maintaining cell viability. GDP-mannose is very important for the correct N-glycosinification process of protein in Arabidopsis and the synthesis of ascorbic acid. cty1 is a mutant with nonsense mutations in GMPase. GMPase is a key enzyme in the synthesis of GDP-mannose. This mutant exhibits an embryonic lethal phenotype. vtc1 and hsn1 are homologous genes of GMPase. Under normal conditions, the phenotypes of these two mutant strains are not different from those of the wild type, but when in a high NH4+ environment, the root length of the mutant strain is obviously suppressed. The embryonic lethality shown in cty1 and the phenotype that the root lengths of vtc1 and hsn1 are sensitive to NH4+ are all caused by the defect of N-glucose activation. However, the question of whether the glycostimulation of protein is related to the process of NH4+ transport across the plasma membrane remains to be further studied.
The harm caused by NH4+ toxicity is great, but we can alleviate it by certain means. Increasing the pH of the solution can alleviate the degree of NH4+ toxicity to a certain extent. In addition, optimizing the light intensity can also alleviate the NH4+ toxicity symptoms of plants grown on the medium with NH4+ as the sole nitrogen source. Because NH4+ poisoning can affect the absorption of nutrient elements by plants, enhancing the concentration of nutrient elements in the environment, especially the concentration of nutrient cations, can also alleviate NH4+ poisoning to a certain extent. Either in the culture medium or in the field, increasing the K+ concentration can alleviate the NH4+ toxicity.
When NH4+ is used as the only nitrogen source, it can form obvious NH4+ poisoning, but when it is used as a nitrogen source with nitrate, the symptoms of NH4+ poisoning can be significantly improved. Moreover, the mixed nitrogen sources can form a kind of synergistic growth, and the total growth rate exceeds the growth of the two nitrogen sources alone. More interestingly, this kind of synergistic growth has also been observed in the growth of conifers. There are two possible explanations for this phenomenon: one is that when nitrogen sources are mixed, it can increase the synthesis of cytokinin; the other is that the absorption of nitrate by plants can cause the alkalinization of plant roots, thereby alleviating the plant roots caused by NH4+ poisoning. The acidification process of the roots.
5 Outlook
For NH4+-sensitive species, excessive NH4+ has a serious inhibitory effect on their growth and yield. Many crops are sensitive to NH4+, and their yield is greatly affected by NH4+ poisoning. In addition, NH4+ poisoning has a significant impact on ecology. A more effective way to alleviate NH4+ toxicity is based on the understanding of NH4+ toxicity mechanism. The use of a mixed medium of nitrate and ammonium nitrogen and increasing the K+ concentration in the culture conditions to alleviate the NH4+ toxicity are successful application examples. However, because the symptoms of NH4+ poisoning are diverse, and it may be related to multiple metabolisms, the mechanism of the poisoning may not be single, which increases the difficulty of studying the NH4+ poisoning mechanism. Considering the degree of harm caused by NH4+ poisoning, more research is needed in the future to better reveal the mechanism of NH4+ poisoning.
Please download “Salt Washing” for countermeasure (the improvement of saline soil can use drainage or salt washing method to eliminate excessive salt)